HOW TO TEST OXYGEN LEVEL IN AIR
HISTORICAL OXYGEN LEVELS
Via Sandy LeonVest; The oxygen crisis; Could the decline of oxygen in the atmosphere undermine our health and threaten human survival?
http://www.theguardian.com/commentisfree/2008/aug/13/carbonemissions.climatechange
And yes it is increasing because of the excessive amount of carbon dioxide it is absorbing. We humans are producing that carbon dioxide with our cars, trucks, planes and energy facilities. But knowing this we can immediately start looking for solutions. We know we must stop the flow of pollutants, heavy metals and thousands of chemicals from the land to the sea. We know we must reduce carbon dioxide emissions. But these changes mean jobs for people. Challenges for people. Yes time is short, but we can do it. Not every species is going to survive the changes.”
TEDxPotomac – Alexandra Cousteau – Connected by Water
The following article forecasts future oxygen depletion and severe repercussions in the world’s oceans, says that reductions in carbon fuel use are needed to prevent this.
http://www.nature.com/ngeo/journal/v2/n2/abs/ngeo420.html
HOW IS OXYGEN MADE?
A simplified overall formula for photosynthesis is:[45]
-
- 6 CO
2 + 6 H
2O + photons → C
6H
12O
6 + 6 O
2
- 6 CO
-
- carbon dioxide + water + sunlight → glucose + dioxygen
Global Oxygen Level Decrease Documented
Scripps Institution of Oceanography (SIO), University of California at San Diego
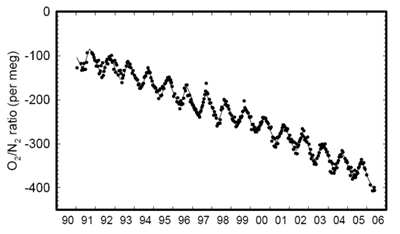
http://www.esrl.noaa.gov/gmd/obop/mlo/programs/coop/scripps/o2/o2.html
Oxygen Bars Are Popular In Polluted Big Cities Like Tokyo And Bejing

Tokyo And Bejing Offer ‘Air Stations’
Why would Tokyo need ‘air stations’ and oxygen bars for people in the city, and have plenty of paying customers, if there was no problem at all with people feeling like they were getting enough oxygen? Is this what the future holds for humanity? Will we all eventually be paying for oxygen in order to feel ‘normal’?
OSHA Enclosed Space Oxygen Requirements
http://www.grainger.com/content/qt-safety-confined-spaces-115
What Happens To A Person Exposed To Lower Oxygen Levels?
Confusion
Cough
Fast heart rate
Rapid breathing
Shortness of breath
Sweating
Wheezing
http://www.webmd.com/asthma/guide/hypoxia-hypoxemia
Hypoxemia explored at Wikipedia
http://en.wikipedia.org/wiki/Hypoxemia
MEASURING OXYGEN LEVEL INSIDE HUMAN BODY
One way to see what the oxygen level is that is actually reaching a person is with a fingertip oxygen
sensor..
“A blood-oxygen monitor displays the percentage of blood that is loaded with oxygen. More specifically, it measures what percentage of hemoglobin, the protein in blood that carries oxygen, is loaded. Acceptable normal ranges for patients without pulmonary pathology are from 95 to 99 percent. For a patient breathing room air at or near sea level, an estimate of arterial pO2 can be made from the blood-oxygen monitor “saturation of peripheral oxygen” (SpO2) reading.”
Pulse oximetry measures solely hemoglobin saturation, not ventilation and is not a complete measure of respiratory sufficiency. It is not a substitute for blood gases checked in a laboratory, because it gives no indication of base deficit, carbon dioxide levels, blood pH, or bicarbonate (HCO3-) concentration. The metabolism of oxygen can be readily measured by monitoring expired CO2, but saturation figures give no information about blood oxygen content. Most of the oxygen in the blood is carried by hemoglobin; in severe anemia, the blood will carry less total oxygen, despite the hemoglobin being 100% saturated.
http://en.wikipedia.org/wiki/Pulse_oximetry
AMOUNT OF OXYGEN AT INCREASING ALTITUDE
This source has the same explanation as above, but is a different source; “If you don’t acclimatise properly, you greatly increase your chance of developing altitude sickness, or even worse, HAPE (high altitude pulmonary oedema) or HACE (high altitude cerebral oedema).
http://www.altitude.org/why_less_oxygen.php
EXAMPLE OF LOCAL MICRO LOW LEVEL OXYGEN LEVEL EVENTS
This is also the reason why you don’t want to stay in a house that is on fire. Oxygen levels can drop rapidly. Get out as fast as you can.
WHAT HAPPENS TO DEEP SEA CREATURES AS CO2 LEVELS KEEP INCREASING?
LOW OXYGEN OCEAN WATER STUDIES
Researchers are discovering that ocean oxygen levels are decreasing. The scientist above discusses low oxygen levels and it’s effect on fish and other sea life.
ONE STUDY POINTS FINGER AT MAN MADE RADIOACTIVE ELEMENTS THAT INCREASE ACID CONTENT OF SEA WATER
WHAT IS CAUSING THE GLOBAL DROP IN OXYGEN LEVELS AND MORE ACIDIC OCEANS?
There are many causes for the drop in oxygen and the rise in CO2 globally, which are explored via the links below.
2014 – List of 45+ Negative Global Tipping Points; via @AGreenRoad
http://agreenroad.blogspot.com/2014/04/2014-list-of-45-global-tipping-points.html
Acid Oceans; Why Should We Care? via @AGreenRoad
http://agreenroad.blogspot.com/2012/06/acid-oceans-why-should-we-care.html
Acid Test; The Global Challenge Of Ocean Acidification; via A Green Road
http://agreenroad.blogspot.com/2012/06/acid-test-global-challenge-of-ocean.html
The Mystery Of Becoming Immortal – Sting Where Is Thy Death? via @AGreenRoad
http://agreenroad.blogspot.com/2013/09/the-mystery-of-becoming-immortal-sting.html
SUMMARY
Humanity needs to focus on reversing this unsustainable negative downward trend and find solutions that will work for 7 future generations, without causing harm. The answers are all here. Click on the link below to access articles that detail solutions to our planetary crisis.
All that is needed is the will and setting a goal to turn this around. It would be a shame to let just a few people (1%) slam the whole planet into a brick wall.
Global Oxygen Levels Are Dropping In Major Cities And Oceans, From A High Of 35% In Geological History; How To Measure Oxygen Levels In Air Yourself
http://agreenroad.blogspot.com/2014/08/global-oxygen-levels-are-dropping-to-as.html
More articles like this one at;
Table of Contents http://tinyurl.com/agrindex 2,000 + Videos And Articles
Access all videos and articles by clicking on Pages – upper left hand corner of any article
Search site – click in SEARCH box – upper right hand corner of any article, type in search word or phrase


Thanks for the information… I really love your blog posts… specially those on Air Oxygen Blender
LikeLiked by 1 person
Thanks for the information… I really love your blog posts… specially those on Baby bassinet
LikeLiked by 1 person
Thanks for the information… I really love your blog posts… specially those on Air Oxygen Blender
LikeLiked by 1 person
Thanks for the information… I really love your blog posts… specially those on Baby incubator
LikeLiked by 1 person
It's kind of crazy to think that this is happening. we really need to reevaluate the way things are being done if we're experiencing an oxygen shortage. This could eventually lead to people needing intensive medical care. Hopefully we can find ways to prevent these issues and increase the amount of oxygen in the air.
http://www.homemedix.com/oxygen-respiratory-sleep.html
LikeLiked by 1 person
I like the site very much. It broads my mind.
TOSHIBA PVM-375AT
LikeLiked by 1 person